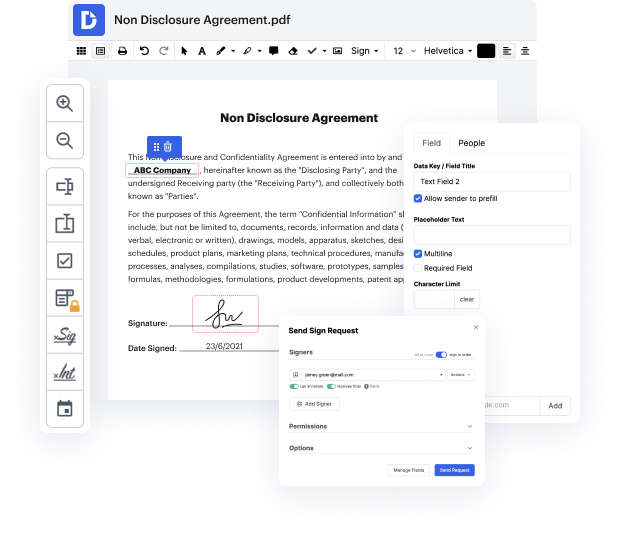
Handling papers like Video Release Consent Letter may seem challenging, especially if you are working with this type for the first time. Sometimes a tiny modification may create a major headache when you don’t know how to work with the formatting and steer clear of making a chaos out of the process. When tasked to add text in Video Release Consent Letter, you can always make use of an image modifying software. Others might go with a classical text editor but get stuck when asked to re-format. With DocHub, though, handling a Video Release Consent Letter is not more difficult than modifying a document in any other format.
Try DocHub for fast and efficient papers editing, regardless of the document format you might have on your hands or the type of document you need to revise. This software solution is online, reachable from any browser with a stable internet connection. Modify your Video Release Consent Letter right when you open it. We’ve developed the interface so that even users without prior experience can readily do everything they require. Simplify your forms editing with one streamlined solution for just about any document type.
Working with different types of documents must not feel like rocket science. To optimize your papers editing time, you need a swift platform like DocHub. Manage more with all our tools on hand.
welcome to the institutional review board for the social and behavioral sciences research education talk on writing an informed consent form at the irb a common question we get from researchers is how do you write a consent form thats why today were going to go over the requirements from the federal regulations and give you tips on writing a consent form the united states department of health and human services has regulations for the protection of human subjects in research the complete regulations can be found at 45 cfr part 46 section 116 but were going to break down and simplify the regulations regarding informed consent here the initial part of the regulations includes a list of concepts as general requirements for informed consent that the researchers should keep in mind and these concepts can be divided into two groups general requirements for informed consent and general requirements for the consent process the first group of general requirements falls under concepts that t