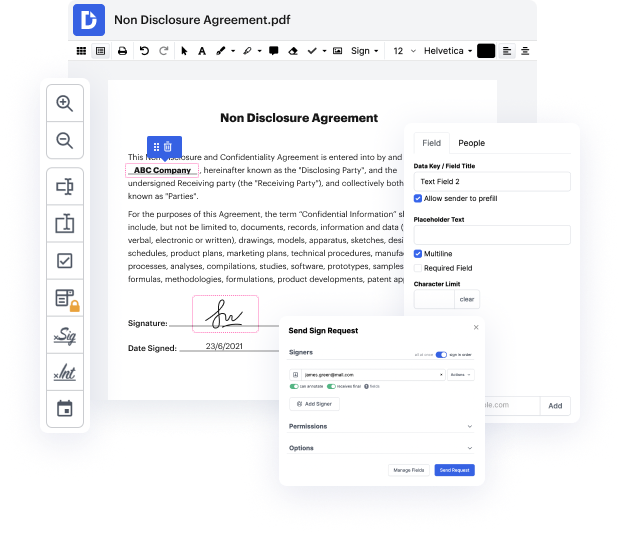
Document generation is a essential element of effective business communication and administration. You need an cost-effective and efficient platform regardless of your document planning point. Quality Incident Record planning can be one of those procedures that require extra care and consideration. Simply stated, you can find greater possibilities than manually creating documents for your small or medium company. Among the best approaches to make sure good quality and efficiency of your contracts and agreements is to set up a multi purpose platform like DocHub.
Editing flexibility is easily the most significant advantage of DocHub. Employ strong multi-use tools to add and take away, or modify any part of Quality Incident Record. Leave comments, highlight information, snip code in Quality Incident Record, and change document managing into an simple and user-friendly process. Gain access to your documents at any time and implement new modifications anytime you need to, which may significantly reduce your time developing exactly the same document from scratch.
Create reusable Templates to streamline your daily routines and steer clear of copy-pasting exactly the same information continuously. Transform, add, and alter them at any moment to ensure you are on the same page with your partners and clients. DocHub can help you prevent errors in often-used documents and provides you with the highest quality forms. Ensure you always keep things professional and remain on brand with your most used documents.
Enjoy loss-free Quality Incident Record modifying and protected document sharing and storage with DocHub. Do not lose any more files or find yourself puzzled or wrong-footed when negotiating agreements and contracts. DocHub enables professionals everywhere to adopt digital transformation as part of their company’s change administration.
so again um as uh i was mentioning um chris anderberg works at sccr as quality and compliance manager um she has over 30 years of research experience working for academic research organizations and industry her knowledge extends to clinical trial management for globe global studies gcp compliance audits regulatory agency inspections clinical event education quality management of clinical trials and policy and procedure development so thank you so much chris for being here with us and i appreciate your time and i hand it over to you you you are muted chris you are muted let me see if i can unmute you there that should be good perfect all right sorry about the technical difficulties ive had these twice this week um thank you very much susan for the very nice introduction im very excited to be here and i wanted to spend our time together talking about quality incident reporting and youll see as we go through were going to be talking about kappas and im hoping that by going through t