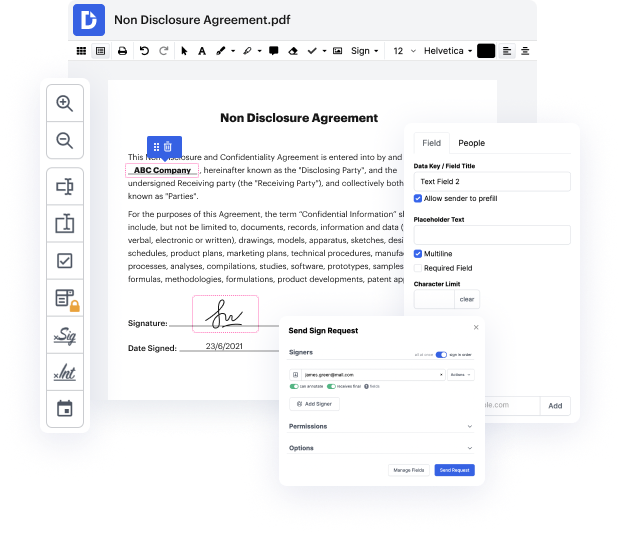
Time is a crucial resource that each business treasures and attempts to change into a benefit. When choosing document management software program, focus on a clutterless and user-friendly interface that empowers consumers. DocHub delivers cutting-edge features to improve your document managing and transforms your PDF file editing into a matter of a single click. Replace Field Validation in the Evaluation Interview Form with DocHub in order to save a ton of efforts and boost your productivity.
Make PDF file editing an easy and intuitive operation that will save you a lot of valuable time. Effortlessly adjust your documents and give them for signing without having switching to third-party software. Give attention to relevant tasks and enhance your document managing with DocHub starting today.
Here are the top 20 Frequently asked Interview Questions about VALIDATION in Pharmaceutical industry. SUBSCRIBE TO CHANNEL FOR MORE INFORMATIVE CONTENT. We will start this series of questions very basic one, What is validation? answer is In simple words it is confidence building procedure for applied process that it will give expected results. USFDA defines it as, The collection and evaluation of data, from the process design stage throughout production, which establishes scientific evidence that a process is capable of consistently delivering quality product. Second question When we should perform validation ? Here are the four cases given when we can perform validation. First one is Before introduction of a new method in use. Second case is Whenever there is change in the conditions for method has been validated e.g. Instrument or equipment with different characteristics. Third case is Whenever the method or process is changed. And forth case is Whenever there is change other th