Document creation is a fundamental aspect of productive business communication and administration. You require an cost-effective and useful platform regardless of your document planning point. Detailed Medical Consent planning can be one of those procedures that need extra care and focus. Simply explained, you can find greater possibilities than manually creating documents for your small or medium business. Among the best approaches to guarantee good quality and usefulness of your contracts and agreements is to set up a multi purpose platform like DocHub.
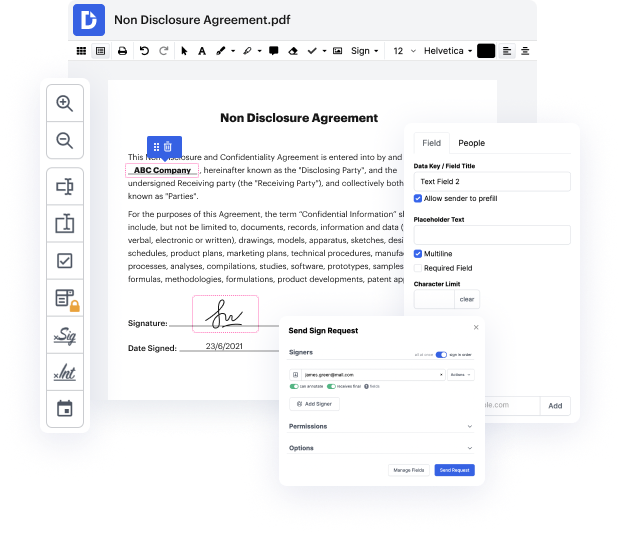
Editing flexibility is the most important advantage of DocHub. Employ robust multi-use instruments to add and remove, or change any element of Detailed Medical Consent. Leave feedback, highlight important information, link page in Detailed Medical Consent, and transform document managing into an simple and intuitive process. Gain access to your documents at any moment and implement new modifications anytime you need to, which could substantially reduce your time making the same document completely from scratch.
Make reusable Templates to make simpler your day-to-day routines and get away from copy-pasting the same details repeatedly. Alter, add, and alter them at any moment to make sure you are on the same page with your partners and customers. DocHub can help you steer clear of mistakes in often-used documents and offers you the highest quality forms. Ensure you keep things professional and remain on brand with the most used documents.
Enjoy loss-free Detailed Medical Consent modifying and secure document sharing and storage with DocHub. Do not lose any more files or find yourself puzzled or wrong-footed when discussing agreements and contracts. DocHub enables specialists anywhere to adopt digital transformation as a part of their company’s change administration.
[Music] today were going to discuss informed consent forms okay um a little bit of what were going to cover its the same type of things right consent informed consent um consent and research who are your audience how does a trial a protocol and a consent time together were going to talk about some of the regulations were going to go through the informed consent process were going to talk about how consents and protocols all fit in to the clinical trial um were going to talk about who your audience is in the case of a clinical study were going to talk about some of the regulations some of the regulations elements of in required elements of informed consent um the dos and donts of writing and informed consent form and then were going to talk about recruitment and how to make sure that you have the the best path forward for assigning subjects up for a study okay lets start off by talking about consent so consent is actually larger than like research and the medical system rig