



Keeping up with the fast-transforming work environment that has appeared since COVID remains to be an essential issue for so many industries. Many organizations search for an accessible and easy-to-implement platform that would be accessible for them 24/7. The principal interest depends on addressing all file generation and approval procedures with minimal time and effort. DocHub provides powerful features and storage integrations that can make positive changes to day-to-day file operations forever. You just need a cost-free DocHub user profile to gain access to Docx Tool for Pharmaceutical Companies | Pharmaceutical Companies Document Management Solution.
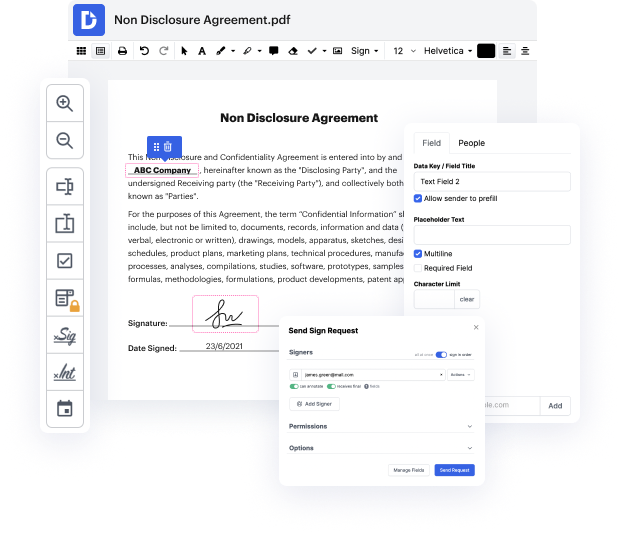
DocHub is an end-to-end editing and annotation platform that handles all your document generation demands. Generate documents completely from scratch, adjust them, leave comments, and quickly gather signatures from your teammates and clients. Forget about losing your documents or worrying for their security - DocHub has industry-leading security standards that safeguard your information.
Focus on agreements and contracts anyplace, at any time. Get the most from Docx Tool for Pharmaceutical Companies | Pharmaceutical Companies Document Management Solution and boost your day-to-day file managing, from file creation to approval and storage.
Pharmaceutical companies need to ensure the safety and effectiveness of their drugs through a powerful quality management system. Qualatize EQMS Suite offers a comprehensive solution for managing quality processes, regulatory compliance, materials, supplier quality, equipment and facilities, controlled documents, employee training, packaging, labeling, customer complaints, and permit/license management. Qualatize helps build a closed-loop quality system to meet regulatory standards and exceed customer expectations, improving product and process quality, minimizing recalls and operational risks, and ensuring compliance with regulatory standards. Built on the salesforce.com platform, Qualatize provides the security, performance, and reliability needed for quality management applications.
At DocHub, your data security is our priority. We follow HIPAA, SOC2, GDPR, and other standards, so you can work on your documents with confidence.
Learn more



