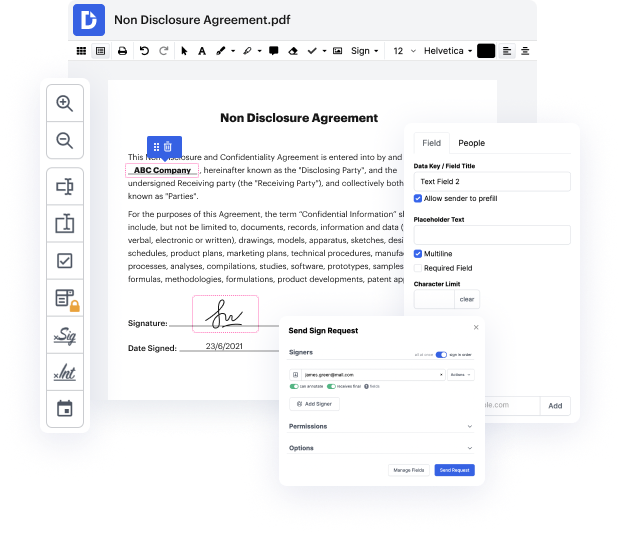
Working with documents means making minor modifications to them everyday. Occasionally, the job goes almost automatically, especially if it is part of your daily routine. Nevertheless, in some cases, dealing with an uncommon document like a Video Release Consent Letter can take valuable working time just to carry out the research. To ensure every operation with your documents is effortless and quick, you need to find an optimal editing solution for this kind of jobs.
With DocHub, you are able to learn how it works without spending time to figure everything out. Your tools are organized before your eyes and are easy to access. This online solution does not require any specific background - training or expertise - from its users. It is ready for work even when you are new to software traditionally utilized to produce Video Release Consent Letter. Quickly create, modify, and share documents, whether you work with them daily or are opening a brand new document type the very first time. It takes moments to find a way to work with Video Release Consent Letter.
With DocHub, there is no need to study different document kinds to figure out how to modify them. Have all the go-to tools for modifying documents close at hand to improve your document management.
This video tutorial from the Institutional Review Board for Social and Behavioral Sciences covers the key points on writing an informed consent form for research. It emphasizes the importance of following federal regulations, specifically 45 CFR part 46 section 116, which outlines the requirements for protecting human subjects. The regulations include general requirements for informed consent and the consent process, which researchers must adhere to when creating consent forms. The video provides tips and guidance to help researchers meet these requirements and ensure compliance with federal regulations.