Document generation and approval certainly are a key priority of each firm. Whether dealing with large bulks of documents or a particular contract, you have to remain at the top of your efficiency. Choosing a ideal online platform that tackles your most typical record creation and approval difficulties could result in a lot of work. Numerous online apps offer you only a minimal set of modifying and signature capabilities, some of which could possibly be useful to manage CCF format. A platform that handles any format and task might be a exceptional choice when choosing program.
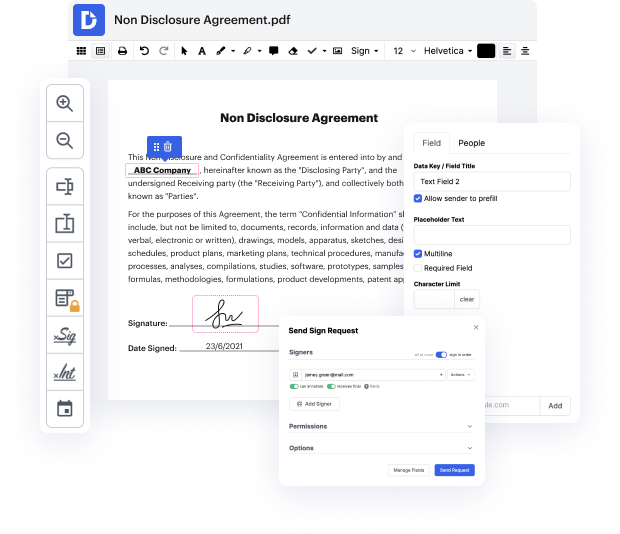
Take file administration and creation to another level of simplicity and sophistication without picking an difficult user interface or expensive subscription options. DocHub gives you tools and features to deal successfully with all of file types, including CCF, and execute tasks of any complexity. Edit, manage, and produce reusable fillable forms without effort. Get full freedom and flexibility to copy impression in CCF anytime and securely store all your complete documents in your user profile or one of many possible integrated cloud storage space apps.
DocHub provides loss-free editing, signature collection, and CCF administration on a expert levels. You do not have to go through tiresome tutorials and spend hours and hours finding out the software. Make top-tier secure file editing a standard process for your every day workflows.
there are several changes to the federal ccf here is what has changed and how you will know the difference between the 2017 ccf and the revised 2020 ccf most of the changes adopted in the revised ccf were made in order to accommodate the use of oral fluid specimen testing that was recently approved for the federal drug testing program now oral fluid drug testing has not yet been authorized for the dot testing program but we do expect guidance to be coming soon for the integration of oral fluid testing into the dot program here are the changes for the revised ccf on all copies one through five step one cdl state and number was added to the donor identification used for fmcsa only on all copies one through five step one the field of other for an example an email address was added to the collector contact info area on copy 1 only step 5a removed the analyte names and check boxes moved the results check boxes and added a line for the docHubing scientist to record the positive analyte and